 
1.) Entropy is defined as the degree of randomness or measure of disorder. 
There is a plenty of reversible process from $A$ to $B$ but we just choose the simplest one for calculations. Yes of course, Entropy is a state function. Where the integral has to be computed through a reversible process. More specifically, the second law of thermodynamics states that as one goes forward in time, the net entropy (degree of. The way we calculate the difference $\Delta S$ is always In the case of entropy, there is some subtlety though. The difference $\Delta S$ between $A$ and $B$ exists even for irreversible paths and it has always the same value. Thus the difference in entropy between the states $A$ and $B$ is simply $\Delta S=S(B)-S(A)$ and this value does not depend on the process that takes $A$ to $B$. The same for the state $B$, which gives $S(B)$. More broadly we define a spontaneous process as one that occurs in the. As discussed previously, we can use this fact to revisit the isotherm + isochore + adiabat circular path (Figure 20.3.1 ). Like enthalpy, entropy is a thermodynamic term that is also a state function. Delta Gibbs free energy (G) is a state function that combines the. The state $A$ has a definite value for the state function entropy, $S(A)$. Because entropy is a state function, it integrates to zero over any circular path going back to initial conditions, just like U and H: d S 0. Both entropy (S) and enthalpy (H) are state functions, while delta entropy (S) is not. We used the statistical definition of entropy and the minimization of the free enthalpy in. Each state has a definite and unique value for the given state function. Entropy S, by the way, is not a state function (TS would be one). This is why entropy is a state function - its value does not depend on the processes required to reach that value. 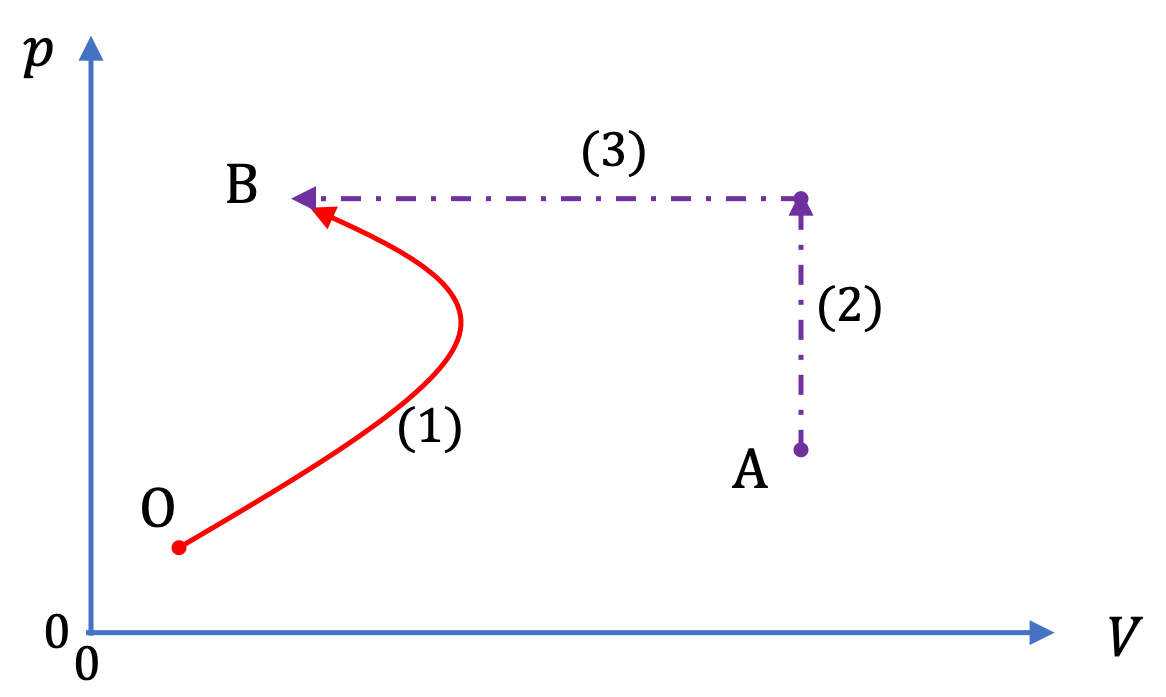
This subtlety is discussed in a bit more detail in a footnote in Kerson Huang's book in page 16.Your question goes right in the kernel of the meaning of the term state function.Ī state function is a function defined over all possible states of the system such that its value for every state does not depend on how the system reached the state. Where $\delta Q_$ by a reversible process. 
In fact, the Clausius statement of the 2nd law says this: We know that the entropy transferred to a system undergoing a reversible cycle is zero, and if you have intuition behind this, then you have intuition behind why entropy is a state function. Now, since $S=k_Bln\Omega$, entropy is also a state function.ĮDIT: I've given some intuition behind the statistical mechanical definition of entropy being a state function, but what about the thermodynamic definition? Therefore $\Omega=\Omega(N,P,T.)$ is a state function. Intuitively, you can think of the number of accessible energy states by particles in a system being influenced by the state of the system (e.g. The number of available energy levels is determined by quantities like temperature, which is also influenced by pressure and other thermodynamic parameters. heating at constant volume and expansion at a constant temperature. Introduction History Classical entropy Statistical entropy v t e Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. Although all forms of energy can be used to do work, it is not possible to use the entire available energy for work. As entropy is a state function, the entropy changes of any process in which temperature and volume both vary is the same as for a path that is divided into two steps i.e. The more disordered a system and higher the entropy, the less of a system's energy is available to do work. Entropy also describes how much energy is not available to do work. A state function is a property depending only on the. The number of microstates is determined by the number of particles (of course) and the number of energy levels available to these particles. Entropy is a measure of the disorder of a system. In classical thermodynamics the entropy is interpreted as a state function of a thermodynamic system. It is represented by S, however it is represented as S in the ordinary state. Entropy is proportional to the number of microstates available to a system, $S=k_Bln\Omega$. Entropy : It is a result of thermodynamics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
